Contents
- Familial pancreatic cancer
- Familial pancreatic cancer causes
- Familial pancreatic cancer signs and symptoms
- Familial pancreatic cancer diagnosis
- Pancreatic cancer staging
- Familial pancreatic cancer treatment
- Pancreatic cancer survival rates
Familial pancreatic cancer
Familial pancreatic cancer is a term to describe families with a high rate of pancreatic cancer or families with a clustering of pancreatic cancer diagnoses. Families are considered to have familial pancreatic cancer if there are at least 2 members of the family with pancreatic cancer who are first-degree relatives, such as a parent, child, or siblings of one another, or if there are at least 3 members of the family who have pancreatic cancer. Healthy individuals who come from a family with familial pancreatic cancer are likely to have an increased risk of developing pancreatic cancer in their lifetime.
At this time, there is no specific test for familial pancreatic cancer. Families are considered to have familial pancreatic cancer if there are:
- 2 or more members of a family who are first-degree relatives, such as parents, children, or siblings, who have been diagnosed with pancreatic cancer, or
- 3 or more close relatives from the same side of the family who have been diagnosed with pancreatic cancer.
If you have symptoms of pancreatic cancer, talk with your doctor. They will perform a physical exam and ask you about your medical history. Your doctor will also recommend specific tests to help find pancreatic cancer.
An estimated 57,600 adults (30,400 men and 27,200 women) in the United States will be diagnosed with pancreatic cancer. About 10% of those cases are thought to be caused by familial pancreatic cancer.
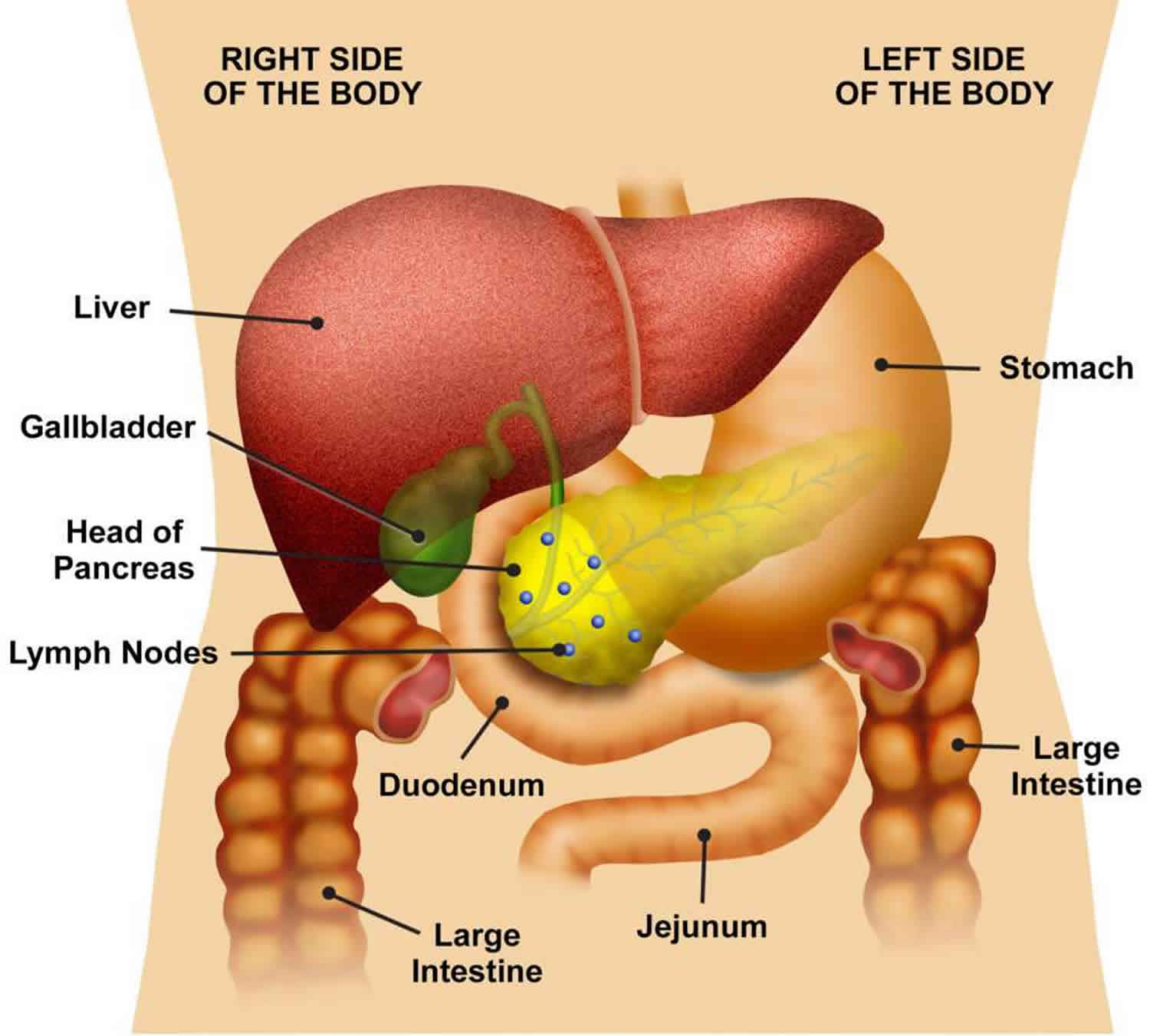
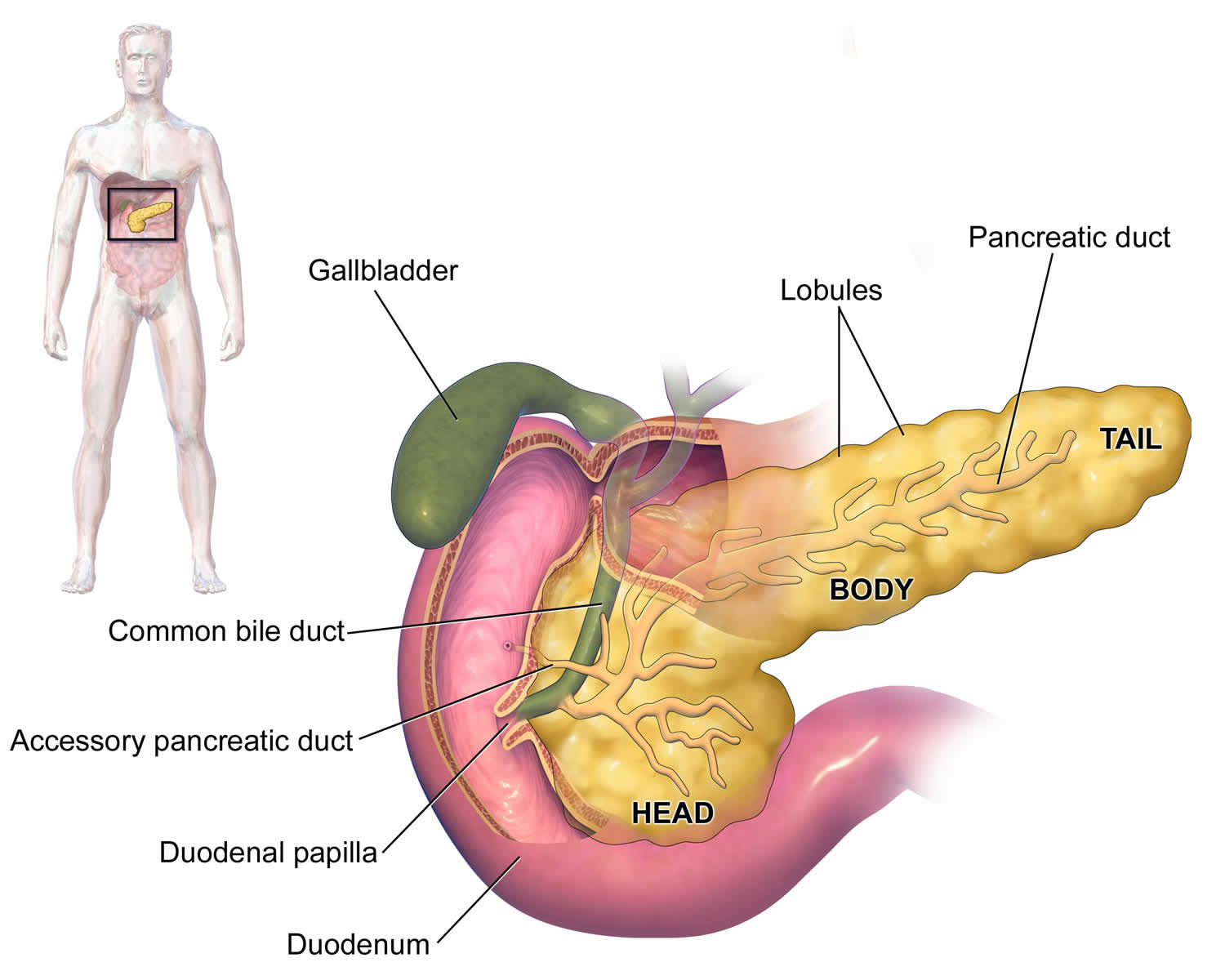
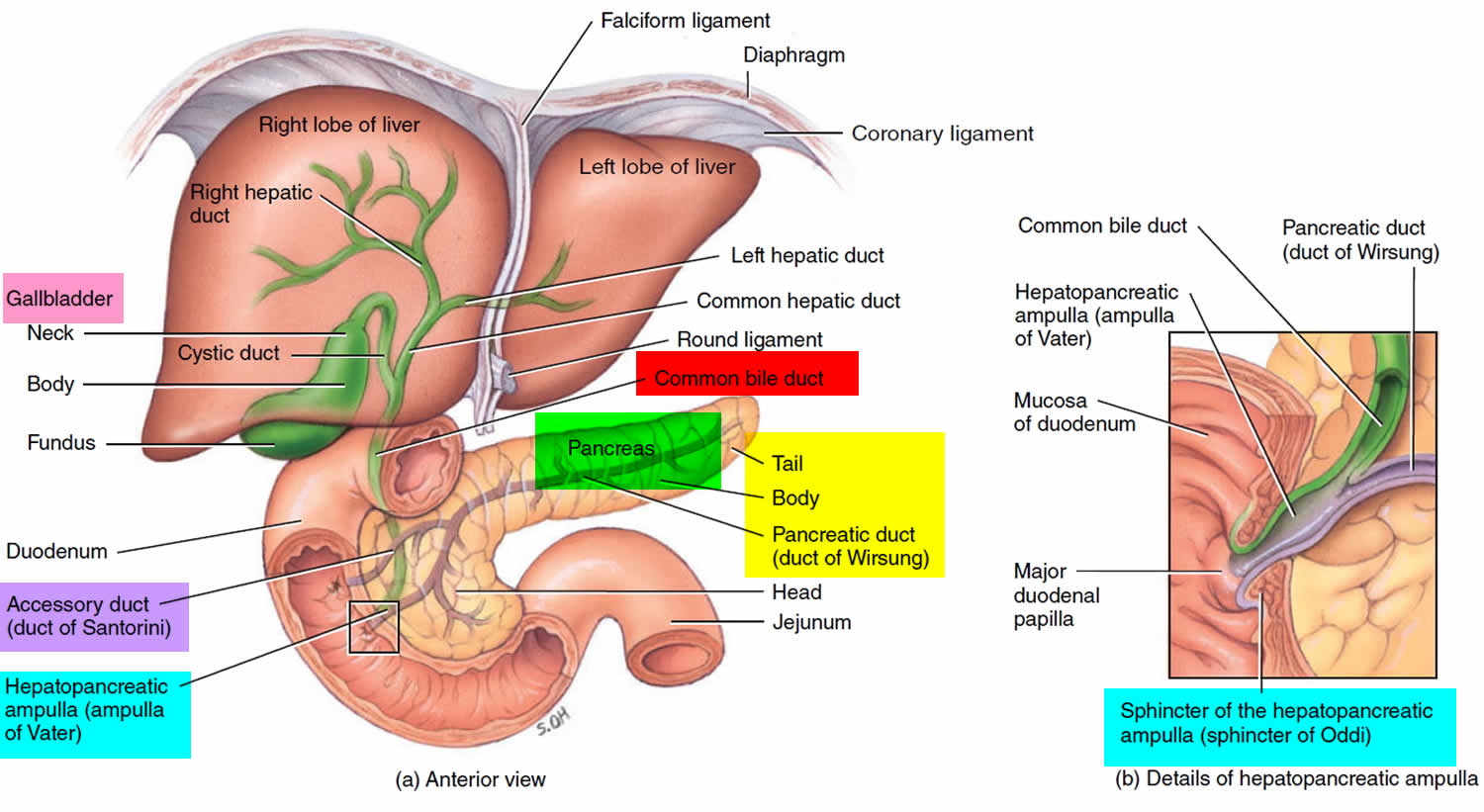
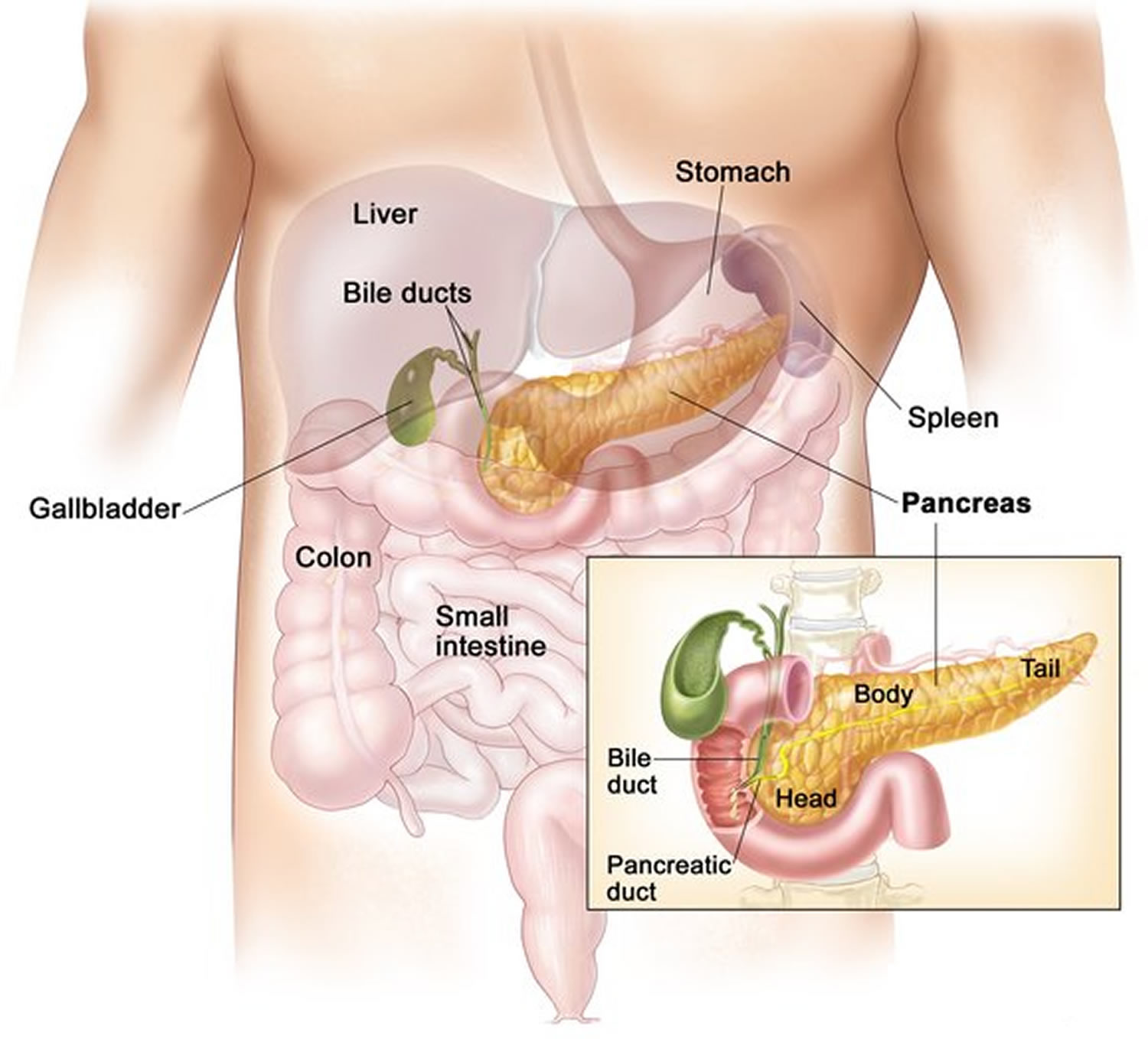
Figure 1. Pancreas anatomy
Figure 2. Pancreas location
Familial pancreatic cancer causes
Individuals from familial pancreatic cancer families should consider genetic testing to see if there is a specific germline genetic mutation that may have caused the pancreatic cancers in their family. A germline mutation is a genetic mutation found in every cell of a person’s body from birth. Some genes linked to familial pancreatic cancer families include BRCA1, BRCA2, PALB2, CDKN2A, and ATM, and the genes linked to Lynch syndrome (MLH1, MSH2, MSH6, PMS2, and EPCAM). Germline mutations in genes that cause other rare inherited cancer syndromes (TP53 mutations in Li-Fraumeni syndrome and STK11 mutations for Peutz-Jeghers syndrome) can also increase the risk of pancreatic cancer. Individuals who carry germline genetic mutations in these genes are at an increased risk of pancreatic cancer as well as other types of cancers. Genetic testing for these genes is available, but your decision to have genetic testing should be discussed carefully with a medical professional with expertise in this area.
It is important to note that genetic testing is still evolving, and only 10% to 20% of families with familial pancreatic cancer will have a mutation identified by genetic testing. Currently, most families with familial pancreatic cancer will have normal genetic testing results, suggesting that the genes responsible for most familial pancreatic cancer families have not yet been discovered. Researchers continue to search for other specific genes that may be linked to familial pancreatic cancer. Since most familial pancreatic cancer families will have normal genetic testing results, it is important to realize that individuals from familial pancreatic cancer families are still at an increased risk of pancreatic cancer, even when genetic testing results are normal. Talk with a genetic counselor before you have any genetic testing.
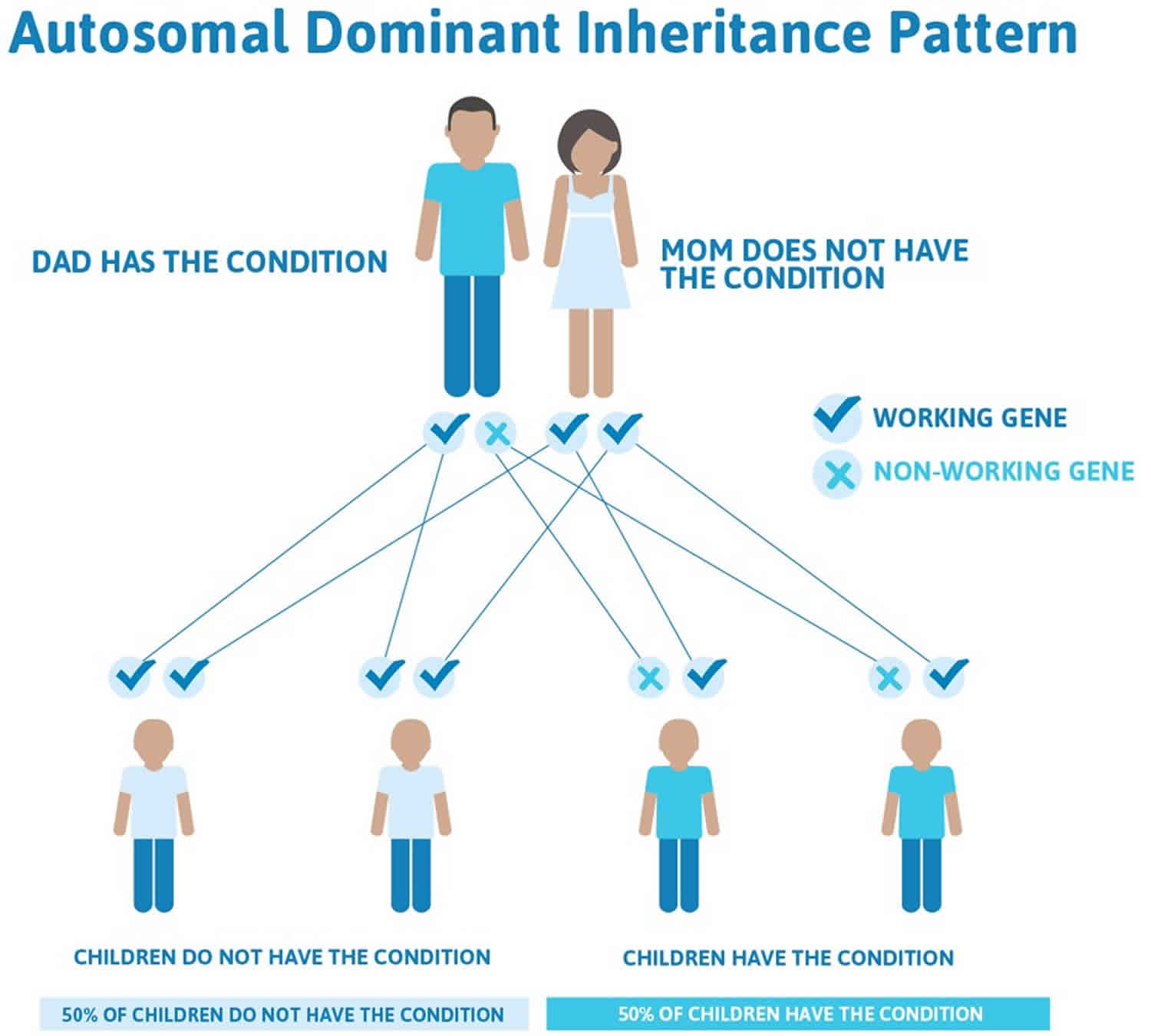
Familial pancreatic cancer inheritance pattern
Normally, every cell has 2 copies of each gene: 1 inherited from the mother and 1 inherited from the father. Researchers think that familial pancreatic cancer typically follows an autosomal dominant inheritance pattern, even though the specific genes that cause familial pancreatic cancer are mostly unknown. In autosomal dominant inheritance, a mutation happens in only 1 copy of the gene. This means that a parent with a gene mutation may pass along a copy of their normal gene. Or, that parent may pass along a copy of the gene with the mutation. Therefore, a child who has a parent with a mutation has a 50% chance of inheriting that mutation. A brother, sister, or parent of a person who has a mutation also has a 50% chance of having the same mutation. However, if the parents test negative for the mutation (meaning each person’s test results found no mutation), the risk to the siblings significantly decreases but their risk may still be higher than an average risk.
Options exist for people interested in having a child when a prospective parent carries a gene mutation that increases the risk for this hereditary cancer syndrome. Preimplantation genetic diagnosis (PGD) is a medical procedure done in conjunction with in-vitro fertilization (IVF). It allows people who carry a specific known genetic mutation to reduce the likelihood that their children will inherit the condition. A woman’s eggs are removed and fertilized in a laboratory. When the embryos reach a certain size, 1 cell is removed and is tested for the hereditary condition in question. The parents can then choose to transfer embryos which do not have the mutation. Preimplantation genetic diagnosis has been in use for over 2 decades and has been used for several hereditary cancer predisposition syndromes. However, this is a complex procedure with financial, physical, and emotional factors to consider before starting. For more information, talk with an assisted reproduction specialist at a fertility clinic.
Figure 3. Familial pancreatic cancer autosomal dominant inheritance pattern
Genetic counseling and testing
If you’ve been diagnosed with pancreatic cancer, your doctor might suggest speaking with a genetic counselor to determine if you could benefit from genetic testing.
Some people with pancreatic cancer have gene mutations (such as BRCA mutations) in all the cells of their body, which put them at increased risk for pancreatic cancer (and possibly other cancers). Testing for these gene mutations can sometimes affect which treatments might be helpful. It might also affect whether other family members should consider genetic counseling and testing as well.
People with specific questions about genetic risks or genetic testing for themselves or family members should speak with a genetics professional.
Resources for locating a genetics professional in your community are available online:
- The National Society of Genetic Counselors (https://www.findageneticcounselor.com/) offers a searchable directory of genetic counselors in the United States and Canada. You can search by location, name, area of practice/specialization, and/or ZIP Code.
- The American Board of Genetic Counseling (https://www.abgc.net/about-genetic-counseling/find-a-certified-counselor/) provides a searchable directory of certified genetic counselors worldwide. You can search by practice area, name, organization, or location.
- The Canadian Association of Genetic Counselors (https://www.cagc-accg.ca/index.php?page=225) has a searchable directory of genetic counselors in Canada. You can search by name, distance from an address, province, or services.
- The American College of Medical Genetics and Genomics (http://www.acmg.net/ACMG/Genetic_Services_Directory_Search.aspx) has a searchable database of medical genetics clinic services in the United States.
What are the estimated cancer risks associated with familial pancreatic cancer?
The lifetime risk of pancreatic cancer for the average individual without a family history of pancreatic cancer is approximately 1%. Individuals with a family history of pancreatic cancer are at an increased lifetime risk for developing pancreatic cancer. This risk is likely higher for individuals from a family with familial pancreatic cancer. The following cancer risk estimates are generalized and should be interpreted with caution since the actual risk for each individual may be different:
- Individuals from familial pancreatic cancer families who have 1 first-degree relative, meaning a parent, sibling, or child, with pancreatic cancer are estimated to have an increased lifetime risk of pancreatic cancer that is 3 to 5 times higher than the general population.
- Individuals from familial pancreatic cancer families who have 2 first-degree relatives with pancreatic cancer are estimated to have an increased lifetime risk of pancreatic cancer that is 5 to 7 times higher than the general population.
- Individuals from familial pancreatic cancer families who have 3 or more first-degree relatives with pancreatic cancer are estimated to have an increased lifetime risk of pancreatic cancer that may be up to 30 times higher than the general population.
Individuals who carry germline mutations in known genes linked to pancreatic cancer risk (BRCA1, BRCA2, PALB2, CDKN2A, ATM, TP53, STK11, MLH1, MSH2, MSH6, PMS2, and EPCAM) are also at an increased risk of various cancers, including pancreatic cancer. For individuals with a mutation in 1 of these genes, the risk of pancreatic cancer may be particularly higher if there is also a history of pancreatic cancer in the family. Recent studies have found that 4% to 10% of individuals with pancreatic cancer will have a mutation in 1 of these genes. Individuals with pancreatic cancer who are of Ashkenazi Jewish ancestry are even more likely to carry 1 of these genetic mutations. Some national guidelines now recommend genetic testing for any person diagnosed with pancreatic cancer, regardless of their family history of cancer or age at diagnosis.
Tobacco use increases an individual’s lifetime risk of pancreatic cancer, regardless of their family history. Tobacco use may significantly increase the risk of pancreatic cancer for individuals from familial pancreatic cancer families.
Familial pancreatic cancer signs and symptoms
Early pancreatic cancers often do not cause any signs or symptoms. By the time they do cause symptoms, they have often grown very large or already spread outside the pancreas.
Having one or more of the symptoms below does not mean you have pancreatic cancer. In fact, many of these symptoms are more likely to be caused by other conditions. Still, if you have any of these symptoms, it’s important to have them checked by a doctor so that the cause can be found and treated, if needed.
Jaundice is yellowing of the eyes and skin. Most people with pancreatic cancer and nearly all people with ampullary cancer will have jaundice as one of their first symptoms.
Jaundice is caused by the buildup of bilirubin, a dark yellow-brown substance made in the liver. Normally, the liver releases a liquid called bile that contains bilirubin. Bile goes through the common bile duct into the intestines, where it helps break down fats. It eventually leaves the body in the stool. When the common bile duct becomes blocked, bile can’t reach the intestines, and the amount of bilirubin in the body builds up.
Cancers that start in the head of the pancreas are near the common bile duct. These cancers can press on the duct and cause jaundice while they are still fairly small, which can sometimes lead to these tumors being found at an early stage. But cancers that start in the body or tail of the pancreas don’t press on the duct until they have spread through the pancreas. By this time, the cancer has often spread beyond the pancreas.
When pancreatic cancer spreads, it often goes to the liver. This can also cause jaundice.
There are other signs of jaundice as well as the yellowing of the eyes and skin:
- Dark urine: Sometimes, the first sign of jaundice is darker urine. As bilirubin levels in the blood increase, the urine becomes brown in color.
- Light-colored or greasy stools: Bilirubin normally helps give stools their brown color. If the bile duct is blocked, stools might be light-colored or gray. Also, if bile and pancreatic enzymes can’t get through to the intestines to help break down fats, the stools can become greasy and might float in the toilet.
- Itchy skin: When bilirubin builds up in the skin, it can start to itch as well as turn yellow.
Pancreatic cancer is not the most common cause of jaundice. Other causes, such as gallstones, hepatitis, and other liver and bile duct diseases, are much more common.
Belly or back pain
Pain in the abdomen (belly) or back is common in pancreatic cancer. Cancers that start in the body or tail of the pancreas can grow fairly large and start to press on other nearby organs, causing pain. The cancer may also spread to the nerves surrounding the pancreas, which often causes back pain. Pain in the abdomen or back is fairly common and is most often caused by something other than pancreatic cancer.
Weight loss and poor appetite
Unintended weight loss is very common in people with pancreatic cancer. These people often have little or no appetite.
Nausea and vomiting
If the cancer presses on the far end of the stomach it can partly block it, making it hard for food to get through. This can cause nausea, vomiting, and pain that tend to be worse after eating.
Gallbladder or liver enlargement
If the cancer blocks the bile duct, bile can build up in the gallbladder, making it larger. Sometimes a doctor can feel this (as a large lump under the right side of the ribcage) during a physical exam. It can also be seen on imaging tests.
Pancreatic cancer can also sometimes enlarge the liver, especially if the cancer has spread there. The doctor might be able to feel the edge of the liver below the right ribcage on an exam, or the large liver might be seen on imaging tests.
Blood clots
Sometimes, the first clue that someone has pancreatic cancer is a blood clot in a large vein, often in the leg. This is called a deep vein thrombosis or DVT. Symptoms can include pain, swelling, redness, and warmth in the affected leg. Sometimes a piece of the clot can break off and travel to the lungs, which might make it hard to breathe or cause chest pain. A blood clot in the lungs is called a pulmonary embolism or PE.
Still, having a blood clot does not usually mean that you have cancer. Most blood clots are caused by other things.
Diabetes
Rarely, pancreatic cancers cause diabetes (high blood sugar) because they destroy the insulin-making cells. Symptoms can include feeling thirsty and hungry, and having to urinate often. More often, cancer can lead to small changes in blood sugar levels that don’t cause symptoms of diabetes but can still be detected with blood tests.
Familial pancreatic cancer diagnosis
If a person has signs and symptoms that might be caused by pancreatic cancer, certain exams and tests will be done to find the cause. If cancer is found, more tests will be done to help determine the extent (stage) of the cancer.
Medical history and physical exam
Your doctor will ask about your medical history to learn more about your symptoms. The doctor might also ask about possible risk factors, including smoking and your family history.
Your doctor will also examine you to look for signs of pancreatic cancer or other health problems. Pancreatic cancers can sometimes cause the liver or gallbladder to swell, which the doctor might be able to feel during the exam. Your skin and the whites of your eyes will also be checked for jaundice (yellowing).
If the results of the exam are abnormal, your doctor will probably order tests to help find the problem. You might also be referred to a gastroenterologist (a doctor who treats digestive system diseases) for further tests and treatment.
Imaging tests
Imaging tests use x-rays, magnetic fields, sound waves, or radioactive substances to create pictures of the inside of your body. Imaging tests might be done for a number of reasons both before and after a diagnosis of pancreatic cancer, including:
- To look for suspicious areas that might be cancer
- To learn how far cancer may have spread
- To help determine if treatment is working
- To look for signs of cancer coming back after treatment
Computed tomography (CT) scan
The CT scan makes detailed cross-sectional images of your body. CT scans are often used to diagnose pancreatic cancer because they can show the pancreas fairly clearly. They can also help show if cancer has spread to organs near the pancreas, as well as to lymph nodes and distant organs. A CT scan can help determine if surgery might be a good treatment option.
If your doctor thinks you might have pancreatic cancer, you might get a special type of CT known as a multiphase CT scan or a pancreatic protocol CT scan. During this test, different sets of CT scans are taken over several minutes after you get an injection of an intravenous (IV) contrast.
CT-guided needle biopsy: CT scans can also be used to guide a biopsy needle into a suspected pancreatic tumor. But if a needle biopsy is needed, most doctors prefer to use endoscopic ultrasound (described below) to guide the needle into place.
Magnetic resonance imaging (MRI)
MRI scans use radio waves and strong magnets instead of x-rays to make detailed images of parts of your body. Most doctors prefer to look at the pancreas with CT scans, but an MRI might also be done.
Special types of MRI scans can also be used in people who might have pancreatic cancer or are at high risk:
- MR cholangiopancreatography (MRCP), which can be used to look at the pancreatic and bile ducts, is described below in the section on cholangiopancreatography.
- MR angiography (MRA), which looks at blood vessels, is mentioned below in the section on angiography.
Ultrasound
Ultrasound (US) tests use sound waves to create images of organs such as the pancreas. The two most commonly used types for pancreatic cancer are:
- Abdominal ultrasound: If it’s not clear what might be causing a person’s abdominal symptoms, this might be the first test done because it is easy to do and it doesn’t expose a person to radiation. But if signs and symptoms are more likely to be caused by pancreatic cancer, a CT scan is often more useful.
- Endoscopic ultrasound (EUS): This test is more accurate than abdominal US and can be very helpful in diagnosing pancreatic cancer. This test is done with a small US probe on the tip of an endoscope, which is a thin, flexible tube that doctors use to look inside the digestive tract and to get biopsy samples of a tumor.
Cholangiopancreatography
This is an imaging test that looks at the pancreatic ducts and bile ducts to see if they are blocked, narrowed, or dilated. These tests can help show if someone might have a pancreatic tumor that is blocking a duct. They can also be used to help plan surgery. The test can be done in different ways, each of which has pros and cons.
Endoscopic retrograde cholangiopancreatography (ERCP): For this test, an endoscope (a thin, flexible tube with a tiny video camera on the end) is passed down the throat, through the esophagus and stomach, and into the first part of the small intestine. The doctor can see through the endoscope to find the ampulla of Vater (where the common bile duct empties into the small intestine).
X-rays taken at this time can show narrowing or blockage in these ducts that might be due to pancreatic cancer. The doctor doing this test can put a small brush through the tube to remove cells for a biopsy or place a stent (small tube) into a bile or pancreatic duct to keep it open if a nearby tumor is pressing on it.
Magnetic resonance cholangiopancreatography (MRCP): This is a non-invasive way to look at the pancreatic and bile ducts using the same type of machine used for standard MRI scans. Unlike ERCP, it does not require an infusion of a contrast dye. Because this test is non-invasive, doctors often use MRCP if the purpose is just to look at the pancreatic and bile ducts. But this test can’t be used to get biopsy samples of tumors or to place stents in ducts.
Percutaneous transhepatic cholangiography (PTC): In this procedure, the doctor puts a thin, hollow needle through the skin of the belly and into a bile duct within the liver. A contrast dye is then injected through the needle, and x-rays are taken as it passes through the bile and pancreatic ducts. As with ERCP, this approach can also be used to take fluid or tissue samples or to place a stent into a duct to help keep it open. Because it is more invasive (and might cause more pain), PTC is not usually used unless ERCP has already been tried or can’t be done for some reason.
Positron emission tomography (PET) scan
For a PET scan, you are injected with a slightly radioactive form of sugar, which collects mainly in cancer cells. A special camera is then used to create a picture of areas of radioactivity in the body.
This test is sometimes used to look for spread from exocrine pancreatic cancers.
PET/CT scan: Special machines can do both a PET and CT scan at the same time. This lets the doctor compare areas of higher radioactivity on the PET scan with the more detailed appearance of that area on the CT scan. This test can help determine the stage (extent) of the cancer. It might be especially useful for spotting cancer that has spread beyond the pancreas and wouldn’t be treatable by surgery.
Angiography
This is an x-ray test that looks at blood vessels. A small amount of contrast dye is injected into an artery to outline the blood vessels, and then x-rays are taken.
An angiogram can show if blood flow in a particular area is blocked by a tumor. It can also show abnormal blood vessels (feeding the cancer) in the area. This test can be useful in finding out if a pancreatic cancer has grown through the walls of certain blood vessels. Mainly, it helps surgeons decide if the cancer can be removed completely without damaging vital blood vessels, and it can also help them plan the operation.
X-ray angiography can be uncomfortable because the doctor has to put a small catheter into the artery leading to the pancreas. Usually the catheter is put into an artery in your inner thigh and threaded up to the pancreas. A local anesthetic is often used to numb the area before inserting the catheter. Once the catheter is in place, the dye is injected to outline all the vessels while the x-rays are being taken.
Angiography can also be done with a CT scanner (CT angiography) or an MRI scanner (MR angiography). These techniques are now used more often because they can give the same information without the need for a catheter in the artery. You might still need an IV line so that a contrast dye can be injected into the bloodstream during the imaging.
Blood tests
Several types of blood tests can be used to help diagnose pancreatic cancer or to help determine treatment options if it is found.
Liver function tests: Jaundice (yellowing of the skin and eyes) is often one of the first signs of pancreatic cancer. Doctors often get blood tests to assess liver function in people with jaundice to help determine its cause. Certain blood tests can look at levels of different kinds of bilirubin (a chemical made by the liver) and can help tell whether a patient’s jaundice is caused by disease in the liver itself or by a blockage of bile flow (from a gallstone, a tumor, or other disease).
Tumor markers: Tumor markers are substances that can sometimes be found in the blood when a person has cancer. Tumor markers that may be helpful in pancreatic cancer are:
- CA 19-9. CA 19-9 is a tumor marker that may be helpful in pancreatic cancer. A drop in the CA 19-9 level after surgery (compared to the level before surgery) and low levels of CA 19-9 after pancreas surgery tend to predict a better prognosis (outlook).
- Carcinoembryonic antigen (CEA), which is not used as often as CA 19-9
Neither of these tumor marker tests is accurate enough to tell for sure if someone has pancreatic cancer. Levels of these tumor markers are not high in all people with pancreatic cancer, and some people who don’t have pancreatic cancer might have high levels of these markers for other reasons. Still, these tests can sometimes be helpful, along with other tests, in figuring out if someone has cancer.
In people already known to have pancreatic cancer and who have high CA19-9 or CEA levels, these levels can be measured over time to help tell how well treatment is working. If all of the cancer has been removed, these tests can also be done to look for signs the cancer may be coming back.
Other blood tests: Other tests, like a complete blood count (CBC) or chemistry panel, can help evaluate a person’s general health (such as kidney and bone marrow function). These tests can help determine if they’ll be able to withstand the stress of a major operation.
Biopsy
A person’s medical history, physical exam, and imaging test results may strongly suggest pancreatic cancer, but usually the only way to be sure is to remove a small sample of tumor and look at it under the microscope. This procedure is called a biopsy. Biopsies can be done in different ways.
Percutaneous (through the skin) biopsy: For this test, a doctor inserts a thin, hollow needle through the skin over the abdomen and into the pancreas to remove a small piece of a tumor. This is known as a fine needle aspiration (FNA). The doctor guides the needle into place using images from ultrasound or CT scans.
Endoscopic biopsy: Doctors can also biopsy a tumor during an endoscopy. The doctor passes an endoscope (a thin, flexible, tube with a small video camera on the end) down the throat and into the small intestine near the pancreas. At this point, the doctor can either use endoscopic ultrasound (EUS) to pass a needle into the tumor or endoscopic retrograde cholangiopancreatography (ERCP) to place a brush to remove cells from the bile or pancreatic ducts.
Surgical biopsy: Surgical biopsies are now done less often than in the past. They can be useful if the surgeon is concerned the cancer has spread beyond the pancreas and wants to look at (and possibly biopsy) other organs in the abdomen. The most common way to do a surgical biopsy is to use laparoscopy (sometimes called keyhole surgery). The surgeon can look at the pancreas and other organs for tumors and take biopsy samples of abnormal areas.
Some people might not need a biopsy
Rarely, the doctor might not do a biopsy on someone who has a tumor in the pancreas if imaging tests show the tumor is very likely to be cancer and if it looks like surgery can remove all of it. Instead, the doctor will proceed with surgery, at which time the tumor cells can be looked at in the lab to confirm the diagnosis. During surgery, if the doctor finds that the cancer has spread too far to be removed completely, only a sample of the cancer may be removed to confirm the diagnosis, and the rest of the planned operation will be stopped.
If treatment (such as chemotherapy or radiation) is planned before surgery, a biopsy is needed first to be sure of the diagnosis.
Lab tests of biopsy samples
The samples obtained during a biopsy (or during surgery) are sent to a lab, where they are looked at under a microscope to see if they contain cancer cells.
If cancer is found, other tests might be done as well. For example, tests might be done to see if the cancer cells have mutations (changes) in certain genes, such as the BRCA genes (BRCA1 or BRCA2) or NTRK genes. This might affect whether certain targeted therapy drugs might be helpful as part of treatment.
Genetic testing for pancreatic cancer risk
It is unknown if screening for pancreatic cancer is effective, and there is no routine screening for pancreatic cancer that is currently recommended for the general population. The medical community continues to research who to screen, which tests to use, and how often to use them.
Given that individuals from familial pancreatic cancer families, or individuals with germline genetic mutations in BRCA1, BRCA2, PALB2, CDKN2A, ATM, MLH1, MSH2, MSH6, PMS2, STK11, and EPCAM, are at increased risk for pancreatic cancer, there is special interest in researching pancreatic cancer screening for these high-risk individuals. It’s important to talk with your doctor about the screening options below, as each person is different.
Current guidelines recommend that healthy individuals from familial pancreatic cancer families should consider pancreatic cancer screening beginning at age 50, or 10 years younger than the earliest pancreatic cancer diagnosis in the family, if at least 1 of the pancreatic cancers in their family was in a first-degree relative. Guidelines also recommend that individuals with germline mutations in the genes listed above should consider screening beginning at age 50, or 10 years younger than the earliest pancreatic cancer diagnosis in the family, if they have a family history of pancreatic cancer. Some experts have recommended that all individuals with germline mutations in STK11 (which causes Peutz-Jeghers syndrome) or CDKN2A (which causes familial atypical multiple mole melanoma [FAMMM] syndrome), have screening regardless of their family history, with Peutz-Jeghers syndrome patients being recommended to begin screening at age 30 to 35 and FAMMM syndrome patients being recommended to begin by age 40. The screening tests that are most commonly used include:
- Magnetic resonance imaging (MRI) – An MRI uses magnetic fields to produce detailed images of the pancreas.
- Endoscopic ultrasound (EUS) – A thin, lighted tube is passed through the patient’s mouth and stomach. The tube goes down into the small intestine to take a picture of the pancreas.
Screening options are likely to change over time as new technologies are developed and more is learned about familial pancreatic cancer. It’s important to talk with your doctor about screening tests that are right for you.
Pancreatic cancer staging
After someone is diagnosed with pancreatic cancer, doctors will try to figure out if it has spread, and if so, how far. This process is called staging. The stage of a cancer describes how much cancer is in the body. It helps determine how serious the cancer is and how best to treat it. Doctors also use a cancer’s stage when talking about survival statistics.
The earliest stage pancreas cancers are stage 0 (carcinoma in situ), and then range from stages I (1) through IV (4). As a rule, the lower the number, the less the cancer has spread. A higher number, such as stage IV, means a more advanced cancer. Cancers with similar stages tend to have a similar outlook and are often treated in much the same way.
The staging system used most often for pancreatic cancer is the AJCC (American Joint Committee on Cancer) TNM system, which is based on 3 key pieces of information:
- The extent of the tumor (T): How large is the tumor and has it grown outside the pancreas into nearby blood vessels?
- The spread to nearby lymph nodes (N): Has the cancer spread to nearby lymph nodes? If so, how many of the lymph nodes have cancer?
- The spread (metastasized) to distant sites (M): Has the cancer spread to distant lymph nodes or distant organs such as the liver, peritoneum (the lining of the abdominal cavity), lungs or bones?
The system described below is the most recent American Joint Committee on Cancer (AJCC) system, effective January 2018. It is used to stage most pancreatic cancers except for well-differentiated pancreatic neuroendocrine tumors (NETs), which have their own staging system.
The staging system in the table uses the pathologic stage. It is determined by examining tissue removed during an operation. This is also known as the surgical stage. Sometimes, if the doctor’s physical exam, imaging, or other tests show the tumor is too large or has spread to nearby organs and cannot be removed by surgery right away or at all, radiation or chemotherapy might be given first. In this case, the cancer will have a clinical stage. It is based on the results of physical exam, biopsy, and imaging tests. The clinical stage can be used to help plan treatment. Sometimes, though, the cancer has spread further than the clinical stage estimates, and may not predict the patient’s outlook as accurately as a pathologic stage.
Numbers or letters after T, N, and M provide more details about each of these factors. Higher numbers mean the cancer is more advanced. Once a person’s T, N, and M categories have been determined, this information is combined in a process called stage grouping to assign an overall stage.
Cancer staging can be complex. If you have any questions about your stage, please ask your doctor to explain it to you in a way you understand.
Table 1. Stages of pancreatic cancer
| AJCC Stage | Stage grouping | Stage description* |
| 0 | Tis
N0 M0 |
The cancer is confined to the top layers of pancreatic duct cells and has not invaded deeper tissues. It has not spread outside of the pancreas. These tumors are sometimes referred to as carcinoma in situ (Tis).
It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| IA | T1
N0 M0 |
The cancer is confined to the pancreas and is no bigger than 2 cm (0.8 inch) across (T1).
It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| IB | T2
N0 M0 |
The cancer is confined to the pancreas and is larger than 2 cm (0.8 inch) but no more than 4cm (1.6 inches) across (T2).
It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| IIA | T3
N0 M0 |
The cancer is confined to the pancreas and is bigger than 4 cm (1.6 inches) across (T3).
It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
|
IIB
|
T1
N1 M0 |
The cancer is confined to the pancreas and is no bigger than 2 cm (0.8 inch) across (T1) AND it has spread to no more than 3 nearby lymph nodes (N1).
It has not spread to distant sites (M0). |
| T2
N1 M0 |
The cancer is confined to the pancreas and is larger than 2 cm (0.8 inch) but no more than 4cm (1.6 inches) across (T2) AND it has spread to no more than 3 nearby lymph nodes (N1).
It has not spread to distant sites (M0). |
|
| T3
N1 M0 |
The cancer is confined to the pancreas and is bigger than 4 cm (1.6 inches) across (T3) AND it has spread to no more than 3 nearby lymph nodes (N1).
It has not spread to distant sites (M0). |
|
| III | T1
N2 M0 |
The cancer is confined to the pancreas and is no bigger than 2 cm (0.8 inch) across (T1) AND it has spread to 4 or more nearby lymph nodes (N2).
It has not spread to distant sites (M0). |
| OR | ||
| T2
N2 M0 |
The cancer is confined to the pancreas and is larger than 2 cm (0.8 inch) but no more than 4cm (1.6 inches) across (T2) AND it has spread to 4 or more nearby lymph nodes (N2).
It has not spread to distant sites (M0). |
|
| OR | ||
| T3
N2 M0 |
The cancer is confined to the pancreas and is bigger than 4 cm (1.6 inches) across (T3) AND it has spread to 4 or more nearby lymph nodes (N2).
It has not spread to distant sites (M0). |
|
| OR | ||
| T4
Any N M0 |
The cancer is growing outside the pancreas and into nearby major blood vessels (T4). The cancer may or may not have spread to nearby lymph nodes (Any N).
It has not spread to distant sites (M0). |
|
| IV | Any T
Any N M1 |
The cancer has spread to distant sites such as the liver, peritoneum (the lining of the abdominal cavity), lungs or bones (M1). It can be any size (Any T) and might or might not have spread to nearby lymph nodes (Any N). |
Footnotes: * The following additional categories are not listed on the table above:
- TX: Main tumor cannot be assessed due to lack of information.
- T0: No evidence of a primary tumor.
- NX: Regional lymph nodes cannot be assessed due to lack of information.
Other prognostic factors
Although not formally part of the TNM system, other factors are also important in determining a person’s prognosis (outlook).
Tumor grade
The grade describes how closely the cancer looks like normal tissue under a microscope.
- Grade 1 (G1) means the cancer looks much like normal pancreas tissue.
- Grade 3 (G3) means the cancer looks very abnormal.
- Grade 2 (G2) falls somewhere in between.
Low-grade cancers (G1) tend to grow and spread more slowly than high-grade (G3) cancers. Most of the time, Grade 3 pancreas cancers tend to have a poor prognosis (outlook) compared to Grade 1 or 2 cancers.
Extent of resection
For patients who have surgery, another important factor is the extent of the resection — whether or not all of the tumor is removed:
- R0: All of the cancer is thought to have been removed. (There are no visible or microscopic signs suggesting that cancer was left behind.)
- R1: All visible tumor was removed, but lab tests of the removed tissue show that some small areas of cancer were probably left behind.
- R2: Some visible tumor could not be removed.
Resectable versus unresectable pancreatic cancer
The AJCC staging system gives a detailed summary of how far the cancer has spread. But for treatment purposes, doctors use a simpler staging system, which divides cancers into groups based on whether or not they can be removed (resected) with surgery:
- Resectable
- Borderline resectable
- Unresectable (either locally advanced or metastatic)
Resectable
If the cancer is only in the pancreas (or has spread just beyond it) and the surgeon believes the entire tumor can be removed, it is called resectable. In general, this would include most stage IA, IB, and IIA cancers in the TNM system.
It’s important to note that some cancers might appear to be resectable based on imaging tests, but once surgery is started it might become clear that not all of the cancer can be removed. If this happens, only some of the cancer may be removed to confirm the diagnosis (if a biopsy hasn’t been done already), and the rest of the planned operation will be stopped to help avoid the risk of major side effects.
Borderline resectable
This term is used to describe some cancers that might have just reached nearby blood vessels, but which the doctors feel might still be removed completely with surgery.
Unresectable
These cancers can’t be removed entirely by surgery.
Locally advanced: If the cancer has not yet spread to distant organs but it still can’t be removed completely with surgery, it is called locally advanced. Often the reason the cancer can’t be removed is because it has grown into or surrounded nearby major blood vessels. (This would include some stage III cancers in the TNM system.)
Surgery to try to remove these tumors would be very unlikely to be helpful and could still have major side effects. Some type of surgery might still be done, but it would be a less extensive operation with the goal of preventing or relieving symptoms or problems like a blocked bile duct or intestinal tract, instead of trying to cure the cancer.
Metastatic: If the cancer has spread to distant organs, it is called metastatic (Stage IV). These cancers can’t be removed completely. Surgery might still be done, but the goal would be to prevent or relieve symptoms, not to try to cure the cancer.
Familial pancreatic cancer treatment
If you’ve been diagnosed with pancreatic cancer, your cancer care team will discuss your treatment options with you. Treatment options and recommendations depend on several factors, including the type and stage of cancer, possible side effects, and the patient’s preferences and overall health.
Depending on the type and stage of the cancer and other factors, treatment options for people with pancreatic cancer can include:
- Surgery
- Ablation or Embolization
- Radiation Therapy
- Chemotherapy
- Targeted Therapy
- Immunotherapy
- Pain Control
- Clinical trials
The doctors on your cancer treatment team might include:
- A surgical oncologist: a doctor who specializes in treating cancer with surgery
- A radiation oncologist: a doctor who specializes in treating cancer with radiation therapy
- A medical oncologist: a doctor who specializes in treating cancer with chemotherapy, immunotherapy, and targeted therapy
- A gastroenterologist: a doctor who specializes in diagnosing and treating diseases of the digestive system.
Many other specialists may be involved in your care as well, including nurse practitioners, nurses, psychologists, social workers, rehabilitation specialists, and other health professionals.
Making treatment decisions
It’s important to discuss all of your treatment options, including their goals and possible side effects, with your doctors to help make the decision that best fits your needs. Some important things to consider include:
- Your age and expected life span
- Any other serious health conditions you have
- The stage (extent) of your cancer
- Whether or not surgery can remove (resect) the cancer
- The likelihood that treatment will cure the cancer (or help in some other way)
- Your feelings about the possible side effects from treatment
You may feel that you must make a decision quickly, but it’s important to give yourself time to absorb the information you have just learned. Ask questions if there is anything you’re not sure about.
If time permits, it is often a good idea to seek a second opinion. A second opinion can give you more information and help you feel more confident about the treatment plan you choose.
Surgery for pancreatic cancer
Two general types of surgery can be used for pancreatic cancer:
- Potentially curative surgery is used when the results of exams and tests suggest that it’s possible to remove (resect) all the cancer.
- Palliative surgery may be done if tests show that the cancer is too widespread to be removed completely. This surgery is done to relieve symptoms or to prevent certain complications like a blocked bile duct or intestine, but the goal is not to cure the cancer.
Staging laparoscopy
To determine which type of surgery might be best, it’s important to know the stage (extent) of the cancer. But it can be hard to stage pancreatic cancer accurately just using imaging tests. Sometimes laparoscopy is done first to help determine the extent of the cancer and if it can be resected.
For this procedure, the surgeon makes a few small incisions (cuts) in the abdomen (belly) and inserts long, thin instruments. One of these has a small video camera on the end so the surgeon can see inside the abdomen and look at the pancreas and other organs. Biopsy samples of tumors and other abnormal areas can show how far the cancer has spread.
Potentially curative surgery
Studies have shown that removing only part of a pancreatic cancer doesn’t help patients live longer, so potentially curative surgery is only done if the surgeon thinks all of the cancer can be removed.
This is a very complex surgery and it can be very hard for patients. It can cause complications and might take weeks or months to recover from fully. If you’re thinking about having this type of surgery, it’s important to weigh the potential benefits and risks carefully.
Fewer than 1 in 5 pancreatic cancers appear to be confined to the pancreas at the time they are found. Even then, not all of these cancers turn out to be truly resectable (able to be completely removed). Sometimes after the surgeon starts the operation it becomes clear that the cancer has grown too far to be completely taken out. If this happens, the operation may be stopped, or the surgeon might continue with a smaller operation with a goal of relieving or preventing symptoms. This is because the planned operation would be very unlikely to cure the cancer and could still lead to major side effects. It would also lengthen the recovery time, which could delay other treatments.
Surgery offers the only realistic chance to cure pancreatic cancer, but it doesn’t always lead to a cure. Even if all visible cancer is removed, often some cancer cells have already spread to other parts of the body. These cells can grow into new tumors over time, which can be hard to treat.
Curative surgery is done mainly to treat cancers in the head of the pancreas. Because these cancers are near the bile duct, they often cause jaundice, which sometimes allows them to be found early enough to be removed completely. Surgeries for other parts of the pancreas are described below, and are done if it’s possible to remove all of the cancer.
Whipple procedure (pancreaticoduodenectomy)
This is the most common operation to remove a cancer in the head of the pancreas.
During this operation, the surgeon removes the head of the pancreas and sometimes the body of the pancreas as well. Nearby structures such as part of the small intestine, part of the bile duct, the gallbladder, lymph nodes near the pancreas, and sometimes part of the stomach are also removed. The remaining bile duct and pancreas are then attached to the small intestine so that bile and digestive enzymes can still go into the small intestine. The end pieces of the small intestine (or the stomach and small intestine) are then reattached so that food can pass through the digestive tract (gut).
Most often, this operation is done through a large incision (cut) down the middle of the belly. Some doctors at major cancer centers also do the operation laparoscopically, which is sometimes known as keyhole surgery.
A Whipple procedure is a very complex operation that requires a surgeon with a lot of skill and experience. It carries a relatively high risk of complications that can be life threatening. When the operation is done in small hospitals or by doctors with less experience, as many as 15% of patients may die as a result of surgical complications. In contrast, when the operation is done in cancer centers by surgeons experienced in the procedure, fewer than 5% of patients die as a direct result of surgery.
To have the best outcome, it’s important to be treated by a surgeon who does many of these operations and to have the surgery at a hospital where many of them are done. In general, people having this type of surgery do better when it’s done at a hospital that does at least 15 to 20 Whipple procedures per year.
Still, even under the best circumstances, many patients have complications from the surgery. These can include:
- Leaking from the various connections between organs that the surgeon has to join
- Infections
- Bleeding
- Trouble with the stomach emptying after eating
- Trouble digesting some foods (which might require taking some pills to help with digestion)
- Weight loss
- Changes in bowel habits
- Diabetes
Distal pancreatectomy
In this operation, the surgeon removes only the tail of the pancreas or the tail and a portion of the body of the pancreas. The spleen is usually removed as well. The spleen helps the body fight infections, so if it’s removed you’ll be at increased risk of infection with certain bacteria. To help with this, doctors recommend that patients get certain vaccines before this surgery.
This surgery is used to treat cancers found in the tail and body of the pancreas. Unfortunately, many of these tumors have usually already spread by the time they are found and surgery is not always an option.
Total pancreatectomy
This operation removes the entire pancreas, as well as the gallbladder, part of the stomach and small intestine, and the spleen. This surgery might be an option if the cancer has spread throughout the pancreas but can still be removed. But this type of surgery is used less often than the other operations because there doesn’t seem to be a major advantage in removing the whole pancreas, and it can have major side effects.
It’s possible to live without a pancreas. But when the entire pancreas is removed, people are left without the cells that make insulin and other hormones that help maintain safe blood sugar levels. These people develop diabetes, which can be hard to manage because they are totally dependent on insulin shots. People who have had this surgery also need to take pancreatic enzyme pills to help them digest certain foods.
Before you have this operation, your doctor will recommend that you get certain vaccines because the spleen will be removed.
Palliative surgery
If the cancer has spread too far to be removed completely, any surgery being considered would be palliative (intended to relieve symptoms). Because pancreatic cancer can spread quickly, most doctors don’t advise major surgery for palliation, especially for people who are in poor health.
Sometimes surgery might be started with the hope it will cure the patient, but once it begins the surgeon discovers this is not possible. In this case, the surgeon might do a less extensive, palliative operation known as bypass surgery to help relieve symptoms.
Cancers growing in the head of the pancreas can block the common bile duct as it passes through this part of the pancreas. This can cause pain and digestive problems because bile can’t get into the intestine. The bile chemicals will also build up in the body, which can cause jaundice, nausea, vomiting, and other problems.
There are two main options to relieve bile duct blockage in this situation:
Stent placement
The most common approach to relieving a blocked bile duct does not involve actual surgery. Instead, a stent (small tube, usually made of metal) is put inside the duct to keep it open. This is usually done through an endoscope (a long, flexible tube) while you are sedated. Often this is part of an endoscopic retrograde cholangiopancreatography (ERCP). The doctor passes the endoscope down the throat and all the way into the small intestine. Through the endoscope, the doctor can then put the stent into the bile duct. The stent can also be put in place through the skin during a percutaneous transhepatic cholangiography (PTC).
The stent helps keep the bile duct open even if the surrounding cancer presses on it. But after several months, the stent may become clogged and may need to be cleared or replaced. Larger stents can also be used to keep parts of the small intestine open if they are in danger of being blocked by the cancer.
A bile duct stent can also be put in to help relieve jaundice before curative surgery is done (which would typically be a couple of weeks later). This can help lower the risk of complications from surgery.
Bypass surgery
In people who are healthy enough, another option for relieving a blocked bile duct is surgery to reroute the flow of bile from the common bile duct directly into the small intestine, bypassing the pancreas. This typically requires a large incision (cut) in the abdomen, and it can take weeks to recover from this. Sometimes surgery can be done through several small cuts in the abdomen using special long surgical tools. This is known as laparoscopic or keyhole surgery.
Having a stent placed is often easier and the recovery is much shorter, which is why this is done more often than bypass surgery. But surgery can have some advantages, such as:
- It can often give longer-lasting relief than a stent, which might need to be cleaned out or replaced.
- It might be an option if a stent can’t be placed for some reason.
- During surgery, the surgeon may be able to cut some of the nerves around the pancreas or inject them with alcohol. Because pancreatic cancer often causes pain if it reaches these nerves, this procedure may reduce or get rid of any pain caused by the cancer.
Sometimes, the end of the stomach is disconnected from the duodenum (the first part of the small intestine) and attached farther down the small intestine during this surgery as well. This is known as a gastric bypass. This is done because over time the cancer might grow large enough to block the duodenum, which can cause pain and vomiting and often requires urgent surgery. Bypassing the duodenum before this happens can sometimes help avoid this.
Bypass surgery can still be a major operation, so it’s important that you are healthy enough to tolerate it and that you talk with your doctor about the possible benefits and risks before you have the surgery.
Ablation or embolization treatments for pancreatic cancer
Ablation and embolization treatments are different ways of destroying tumors, rather than removing them with surgery. They are used much less often for pancreatic cancers but can sometimes be used to help treat pancreatic cancer that has spread to other organs, especially the liver.
These treatments are very unlikely to cure cancers on their own. They are more likely to be used to help prevent or relieve symptoms, when there are only a few areas of spread, and are often used along with other types of treatment.
Ablative treatments
Ablation refers to treatments that destroy tumors, usually with extreme heat or cold. They are generally best for tumors no more than about 2 cm (a little less than an inch) across. Typically, with this type of treatment you will not need to stay in the hospital. There are different kinds of ablative treatments:
Radiofrequency ablation (RFA) uses high-energy radio waves for treatment. A thin, needle-like probe is put through the skin and into the tumor. Placement of the probe is guided by ultrasound or CT scans. The tip of the probe releases a high-frequency electric current which heats the tumor and destroys the cancer cells.
Microwave thermotherapy is similar to radiofrequency ablation, except it uses microwaves to heat and destroy the cancer cells.
Ethanol (alcohol) ablation also known as percutaneous ethanol injection kills the cancer cells by injecting concentrated alcohol directly into the tumor. This is usually done through the skin using a needle guided by ultrasound or CT scans.
Cryosurgery also known as cryotherapy or cryoablation destroys a tumor by freezing it with a thin metal probe. The probe is guided through the skin and into the tumor, using ultrasound. Then very cold gasses are passed through the probe to freeze the tumor, killing the cancer cells. This method may be used to treat larger tumors than the other ablation techniques, but it sometimes requires general anesthesia (where you are put into a deep sleep).
Side effects of ablation treatments
Possible side effects after ablation therapy include abdominal pain, infection, and bleeding inside the body. Serious complications are uncommon, but they are possible.
Embolization
During embolization, substances are injected into an artery to try to block the blood flow to cancer cells, causing them to die. This may be used for larger tumors (up to about 5 cm or 2 inches across) in the liver.
There are 3 main types of embolization:
- Arterial embolization also known as trans-arterial embolization (TAE) involves putting a catheter (a thin, flexible tube) into an artery through a small cut in the inner thigh and threaded up into the hepatic artery feeding the tumor. Blood flow is blocked (or reduced) by injecting materials that plug up that artery. Most of the healthy liver cells will not be affected because they get their blood supply from a different blood vessel, the portal vein.
- Chemoembolization also known as trans-arterial chemoembolization (TACE) combines embolization with chemotherapy. Most often, this is done by using tiny beads that give off a chemotherapy drug during the embolization. TACE can also be done by giving chemotherapy through the catheter directly into the artery, then plugging up the artery.
- Radioembolization combines embolization with radiation therapy. In the United States, this is done by injecting small radioactive beads (called microspheres) into the hepatic artery. The beads lodge in the blood vessels near the tumor, where they give off small amounts of radiation to the tumor site. Since the radiation travels a very short distance, its effects are limited mainly to the tumor.
Side effects of embolization
Possible side effects after embolization include abdominal pain, fever, nausea, infection, and blood clots in nearby blood vessels. Serious complications are not common, but they can happen.
Radiation therapy for pancreatic cancer
Radiation therapy uses high-energy x-rays (or particles) to kill cancer cells. It can be helpful in treating some pancreatic cancers.
Radiation therapy might be used when:
- Radiation might be given after surgery (known as adjuvant treatment) to try to lower the chance of the cancer coming back. The radiation is typically given along with chemotherapy, which is together known as chemoradiation or chemoradiotherapy.
- For borderline resectable tumors, radiation might be given along with chemotherapy before surgery (neoadjuvant treatment) to try to shrink the tumor and make it easier to remove completely.
- Radiation therapy combined with chemotherapy may be used as part of the main treatment in people whose cancers have grown beyond the pancreas and can’t be removed by surgery (locally advanced/unresectable cancers).
- Radiation is sometimes used to help relieve symptoms (such as pain) in people with advanced cancers or in people who aren’t healthy enough for other treatments like surgery.
The type of radiation most often used to treat pancreatic cancer known as external beam radiation therapy focuses radiation from a source outside of the body on the cancer.
Getting radiation therapy is much like getting an x-ray, but the radiation is stronger. The procedure itself is painless. Each treatment lasts only a few minutes, although the setup time – getting you into place for treatment – usually takes longer. Most often, radiation treatments are given 5 days a week for several weeks.
Possible side effects of radiation therapy
Some of the more common side effects of radiation therapy include:
- Skin changes in areas getting radiation, ranging from redness to blistering and peeling
- Nausea and vomiting
- Diarrhea
- Fatigue
- Loss of appetite
- Weight loss
Radiation can also lower blood counts, which can increase the risk of serious infection.
Usually these effects go away within a few weeks after the treatment is complete. Ask your doctor what side effects to expect and how to prevent or relieve them.
Chemotherapy for pancreatic cancer
Chemotherapy (chemo) is an anti-cancer drug injected into a vein or taken by mouth. These drugs enter the bloodstream and reach almost all areas of the body, making this treatment potentially useful for cancers whether or not they have spread.
Chemo is often part of the treatment for pancreatic cancer and may be used at any stage:
- Before surgery (neoadjuvant chemotherapy): Chemo can be given before surgery (sometimes along with radiation) to try to shrink the tumor so it can be removed with less extensive surgery. Neoadjuvant chemo is often used to treat cancers that are too big to be removed by surgery at the time of diagnosis (called locally advanced cancers).
- After surgery (adjuvant chemotherapy): Chemo can be used after surgery (sometimes along with radiation) to try to kill any cancer cells that have been left behind or have spread but can’t be seen, even on imaging tests. If these cells were allowed to grow, they could form new tumors in other places in the body. This type of treatment might lower the chance that the cancer will come back later.
- For advanced pancreatic cancer: Chemo can be used when the cancer is advanced and can’t be removed completely with surgery, or if surgery isn’t an option, or if the cancer has spread to other organs.
When chemo is given along with radiation, it is known as chemoradiation. It helps the radiation work better, but can also have more side effects.
Chemo drugs are used for pancreatic cancer
In most cases (especially as adjuvant or neoadjuvant treatment), chemo is most effective when combinations of drugs are used. For people who are healthy enough, 2 or more drugs are usually given together. For people who are not healthy enough for combined treatments, a single drug (usually gemcitabine, 5-FU, or capecitabine) can be used.
The most common drugs used for adjuvant and neoadjuvant chemo include:
- Gemcitabine (Gemzar)
- 5-fluorouracil (5-FU)
- Oxaliplatin (Eloxatin)
- Albumin-bound paclitaxel (Abraxane)
- Capecitabine (Xeloda)
- Cisplatin
- Irinotecan (Camptosar)
Chemotherapy for advanced pancreatic cancer
- Gemcitabine (Gemzar)
- 5-fluorouracil (5-FU) or Capecitabine (Xeloda) (an oral 5FU drug)
- Irinotecan (Camptosar) or Liposomal Irinotecan (Onivyde)
- Platinum agents : Cisplatin and Oxaliplatin (Eloxatin)
- Taxanes: Paclitaxel (Taxol), Docetaxel (Taxotere), and Albumin-bound paclitaxel (Abraxane)
How chemotherapy is given for pancreatic cancer
Chemo drugs for pancreatic cancer can be given into a vein (IV) or by mouth as a pill. The infusion can be done in a doctor’s office, chemotherapy clinic, or in a hospital setting.
Often, a slightly larger and sturdier IV is required in the vein system to give chemo. They are known as central venous catheters (CVCs), central venous access devices (CVADs), or central lines. They are used to put medicines, blood products, nutrients, or fluids right into your blood. They can also be used to take out blood for testing.
Doctors give chemo in cycles, with each period of treatment followed by a rest period to give you time to recover from the effects of the drugs. Cycles are most often 2 or 3 weeks long. The schedule varies depending on the drugs used. For example, with some drugs, the chemo is given only on the first day of the cycle. With others, it is given for a few days in a row, or once a week. Then, at the end of the cycle, the chemo schedule repeats to start the next cycle.
Adjuvant and neoadjuvant chemo is often given for a total of 3 to 6 months, depending on the drugs used. The length of treatment for advanced pancreatic cancer is based on how well it is working and what side effects you may have.
Side effects of chemotherapy for pancreatic cancer
Chemo drugs can cause side effects. These depend on the type and dose of drugs given and how long treatment lasts. Common possible side effects include:
- Nausea and vomiting
- Loss of appetite
- Hair loss
- Mouth sores
- Diarrhea or constipation
Chemo can also affect the blood-forming cells of the bone marrow, which can lead to:
- Increased chance of infection (from low white blood cells)
- Bleeding or bruising (from low platelet counts)
- Fatigue or shortness of breath (from low red blood cells)
These side effects usually go away after treatment is finished. There are often ways to lessen these side effects. For example, drugs can be given to help prevent or reduce nausea and vomiting.
Some chemo drugs can cause other side effects. For example:
- Drugs such as cisplatin, oxaliplatin, and paclitaxel can damage nerves, which can lead to symptoms of numbness, tingling, or even pain in the hands and feet (called peripheral neuropathy). For a day or so after treatment, oxaliplatin can cause nerve pain that gets worse with exposure to cold, including when swallowing cold foods or liquids.
- Cisplatin can damage the kidneys. Doctors try to prevent this by giving the patient lots of intravenous (IV) fluid before and after the drug is given.
- Cisplatin can affect hearing. Your doctor may ask if you have any ringing in the ears or hearing loss during treatment.
Targeted therapy for pancreatic cancer
As researchers have learned more about the changes in pancreatic cancer cells that help them grow, they have developed newer drugs to specifically target these changes. These targeted drugs work differently from standard chemo drugs. Sometimes they work when standard chemo drugs don’t, and they often have different side effects.
EGFR inhibitor
Erlotinib (Tarceva) is a drug that targets a protein on cancer cells called EGFR, which normally helps the cells grow. In people with advanced pancreatic cancer, this drug can be given along with the chemo drug gemcitabine. Some people may benefit more from this combination than others.
This drug is taken as a pill, once a day.
Common side effects of erlotinib include an acne-like rash on the face and neck, diarrhea, loss of appetite, and feeling tired. Less common but more serious side effects can include serious lung, liver, or kidney damage; holes (perforations) forming in the stomach or intestines; serious skin conditions; and bleeding or blood clotting problems.
PARP inhibitor
In a small number of pancreatic cancers, the cells have changes in one of the BRCA genes (BRCA1 or BRCA2). Changes in one of these genes can sometimes lead to cancer.
Olaparib (Lynparza) is a type of drug known as a PARP inhibitor. PARP enzymes are normally involved in a pathway that helps repair damaged DNA inside cells. The BRCA genes are normally involved in a different pathway of DNA repair, and mutations in one of these genes can block that pathway. By blocking the PARP pathway as well, this drug makes it very hard for tumor cells with a mutated BRCA gene to repair damaged DNA, which often leads to their death.
Olaparib can be used to treat advanced pancreatic cancer in people with a known or suspected BRCA gene mutation, whose cancer has not gotten worse after at least 4 months of chemo that included a platinum drug (such as oxaliplatin or cisplatin).
This drug has been shown to help shrink or slow the growth of some advanced pancreatic cancers, although so far it’s not clear if it can help people live longer.
This drug is taken by mouth as pills, typically twice a day.
Side effects of this drug can include nausea, vomiting, diarrhea or constipation, fatigue, feeling dizzy, loss of appetite, taste changes, low red blood cell counts (anemia), low white blood cell counts (with an increased risk of infection), belly pain, and muscle and joint pain. Less common but more serious side effects can include inflammation in the lungs and the development of certain blood cancers, such as myelodysplastic syndrome (MDS) or acute myeloid leukemia (AML).
NTRK inhibitors
A small number of pancreatic cancers have changes in one of the NTRK genes. These gene changes can sometimes lead to abnormal cell growth and cancer.
Larotrectinib (Vitrakvi) and entrectinib (Rozlytrek) target the proteins made by the NTRK genes. These drugs can be used in people with advanced pancreatic cancer that has been found to have an NTRK gene change, typically when the cancer is still growing despite other treatments.
These drugs are taken as pills, once or twice daily.
Common side effects of these drugs can include dizziness, fatigue, nausea, vomiting, constipation, weight gain, and diarrhea. Less common but more serious side effects can include abnormal liver tests, heart problems, and confusion.
Immunotherapy for pancreatic cancer
Immunotherapy is the use of medicines to stimulate a person’s own immune system to recognize and destroy cancer cells more effectively. Certain types of immunotherapy can be used to treat pancreatic cancer.
Immune checkpoint inhibitors
An important part of the immune system is its ability to keep itself from attacking the body’s normal cells. To do this, it uses “checkpoint” proteins on immune cells, which act like switches that need to be turned on (or off) to start an immune response. Cancer cells sometimes use these checkpoints to keep the immune system from attacking them. But drugs that target these checkpoints hold a lot of promise as cancer treatments.
Drugs called checkpoint inhibitors can be used for people whose pancreatic cancer cells have tested positive for specific gene changes, such as a high level of microsatellite instability (MSI-H), or changes in one of the mismatch repair (MMR) genes. Changes in MSI or in MMR genes (or both) are often seen in people with Lynch syndrome.
The drugs are used for people whose cancer starts growing again after chemotherapy. They might also be used to treat people whose cancer can’t be removed with surgery, has come back (recurred) after treatment, or has spread to other parts of the body (metastasized).
PD-1 inhibitor
Pembrolizumab (Keytruda) is a drug that targets PD-1, a checkpoint protein on immune system cells called T cells, that normally helps keep these cells from attacking normal cells in the body. By blocking PD-1, this drug boosts the immune response against pancreatic cancer cells and can often shrink tumors.
This drug is given as an intravenous (IV) infusion every 2 or 3 weeks.
Side effects can include fatigue, cough, nausea, itching, skin rash, decreased appetite, constipation, joint pain, and diarrhea.
Other, more serious side effects occur less often. This drug works by basically removing the brakes from the body’s immune system. Sometimes the immune system starts attacking other parts of the body, which can cause serious or even life-threatening problems in the lungs, intestines, liver, hormone-making glands, kidneys, or other organs.
It’s very important to report any new side effects to your health care team promptly. If serious side effects do occur, treatment may need to be stopped and you may get high doses of corticosteroids to suppress your immune system.
Pain control for pancreatic cancer
Pain can be a major problem for people with pancreatic cancer. These cancers can invade and press on nerves near the pancreas, which can cause pain in the abdomen (belly) or back.
Treatment is available to help relieve this pain. If you are having any pain, please be sure to tell your doctor or nurse. Pain is easier to control if the treatment is started when you first have it. You and your doctor or nurse can talk about the best ways to treat your pain. A pain specialist can also help develop a treatment plan.
Some proven ways to relieve pain from pancreatic cancer include:
Pain medicines
For most patients, morphine or similar drugs (opioids) can help control the pain. Many people are worried about these drugs because they fear becoming addicted, but studies have shown that the risk of this is low if the patient takes the drug for pain as directed by the doctor.
Pain medicines work best when they are taken on a regular schedule. They do not work as well if they are only used when the pain becomes severe. Several long-acting forms of morphine and other opioids are in pill form and only need be taken once or twice a day. There is even a long-acting form of the drug fentanyl that is applied as a patch every 3 days.
Common side effects of these drugs are nausea and feeling sleepy, which often get better over time. Constipation is a common side effect that does not get better on its own, so it needs to be treated. Most people need to take stool softeners and/or laxatives daily.
Other treatments
Sometimes certain procedures might be needed to treat pain. For example, cutting or injecting alcohol into some of the nerves (that carry pain sensations) near the pancreas can often improve pain and may allow you to use lower doses of pain medicines. If you are having surgery for some reason (such as to remove the cancer or relieve bile duct blockage), this can usually be done as part of the same operation.
This can also be done as a separate procedure. The doctor might do a nerve block by injecting the nerves near the pancreas with either an anesthetic or a medicine that destroys the nerves.
This can be done with the help of an ultrasound or CT scan either by:
- passing a needle through the skin or
- by using an endoscope (a long, flexible tube that is passed down the throat and past the stomach) that guides a needle to the nerves.
Treating the cancer with chemotherapy and/or radiation therapy can also sometimes relieve pain by shrinking the size of the cancer.
Pancreatic cancer survival rates
Survival rates can give you an idea of what percentage of people with the same type and stage of cancer are still alive a certain amount of time (usually 5 years) after they were diagnosed. They can’t tell you how long you will live, but they may help give you a better understanding of how likely it is that your treatment will be successful.
Keep in mind that survival rates are estimates and are often based on previous outcomes of large numbers of people who had a specific cancer, but they can’t predict what will happen in any particular person’s case. These statistics can be confusing and may lead you to have more questions. Talk with your doctor about how these numbers may apply to you, as he or she is familiar with your situation.
A relative survival rate compares people with the same type and stage of pancreatic cancer to people in the overall population. For example, if the 5-year relative survival rate for a specific stage of pancreatic cancer is 50%, it means that people who have that cancer are, on average, about 50% as likely as people who don’t have that cancer to live for at least 5 years after being diagnosed.
Table 2. 5-year relative survival rates for pancreatic cancer (based on people diagnosed with pancreatic cancer between 2009 and 2015)
| Surveillance, Epidemiology, and End Results Stage | 5-year Relative Survival Rate |
| Localized | 37% |
| Regional | 12% |
| Distant | 3% |
| All SEER stages combined | 9% |
Footnotes:
The Surveillance, Epidemiology, and End Results (SEER) database does not group cancers by AJCC TNM stages (stage 1, stage 2, stage 3, etc.). Instead, it groups cancers into localized, regional, and distant stages:
- Localized: There is no sign that the cancer has spread outside of the pancreas.
- Regional: The cancer has spread from the pancreas to nearby structures or lymph nodes.
- Distant: The cancer has spread to distant parts of the body such as the lungs, liver or bones.
- These numbers apply only to the stage of the cancer when it is first diagnosed. They do not apply later on if the cancer grows, spreads, or comes back after treatment.
- These numbers don’t take everything into account. Survival rates are grouped based on how far the cancer has spread, but your age, overall health, how well the cancer responds to treatment, tumor grade, extent of resection, level of tumor marker (CA 19-9) and other factors will also affect your outlook.
- People now being diagnosed with pancreatic cancer may have a better outlook than these numbers show. Treatments improve over time, and these numbers are based on people who were diagnosed and treated at least five years earlier.